
Now featuring ALLClear™ Technology Just Load and GO!
ALLClear™ Technology is designed to ensure optimal package and instrument conditions for sterilization, and provide:
- Instrument Warming
- Moisture Detection & Correction of Load
- System Diagnostic Check Prior to Sterilizaton Cycle

CONNECT. CORRELATE. COMMUNICATE
ASP ACCESS™ is ASP’s smart information-sharing technology providing data collection and communication of results across ASP sterilizers and hospital networks connected to ASP ACCESS™ Technology. Learn more about ASP ACCESS™
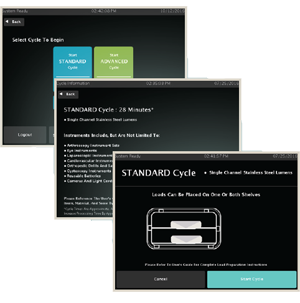
COMPLIANCE IS CRUCIAL
Simple, easy-to-use system that reduces operator error
Graphic display indicating proper load placement for the selected cycle
Cycle information screens detailing the types of devices that can be processed in each cycle
Intuitive, on-screen instructions simplify system operation and staff training

FLEXIBILITY & SPEED
- Process instrument sets in as little as 28 minutes, or 38 minutes for single-channel flexible endoscopes.
- Upgradable System that enhances processing capability.
- Networking capability with multiple instrument tracking systems and hospital servers.
- STERRAD® Systems have over 23,000 medical device manufacturer endorsements and available on our STERRAD Sterility Guide. Click here to learn more

Designed for Safety by a Trusted Partner
Safety is a primary concern of ASP. There are over 15 million cycles successfully completed worldwide in 20,000 STERRAD® System.These cycles use a combination of hydrogen peroxide and plasma to rapidly sterilize medical instrumentation without leaving toxic residues and have been shown in a recent study* to significantly reduce worker exposure to hydrogen peroxide emissions compared to STERIS V-PRO®. Click here to learn more.
The system also includes:
- Touch free sterilant cassette removal
- Quality control integration
- Hydrogen peroxide monitor
*Comparison Study of Environmental Hydrogen Peroxide Levels of STERRAD® Systems and STERIS V-PRO® Low Temperature Sterilizers Reveal Striking Differences. The research was designed and executed by Actionable Research, an independent third-party research firm in conjunction with ChemDAQ® Inc., a manufacturer of environmental safety monitoring systems. The research sponsor was Advanced Sterilization Products. All data were collected by the ChemDAQ® staff.








 Terminal Sterilization
Terminal Sterilization


